The most common form of hearing loss worldwide is difficulty understanding words in the presence of background sound or noise. Sometimes termed “hidden hearing loss”, this is a problem that is difficult to diagnose because the standard hearing test (audiogram) cannot measure this type of hearing loss. There is currently no FDA-approved drug or surgery for treatment of this hearing problem. During the past 10 years, we have gained insight into the causes and possible remedies for this issue. According to research published in the July 14, 2020 issue of Frontiers in Molecular Neuroscience, animal studies strongly suggest that a currently available drug used for a completely unrelated condition may be repurposed for improving age- and noise-related hearing loss and speech comprehension in noisy environments
What is at issue here?
The World Health Organization (WHO) and other sources estimate hearing loss affects 466 million people worldwide and will affect almost a billion people by 2050. The cost of unaddressed hearing loss is estimated at $750 billion/year globally. Most of this burden is due to sensorineural hearing loss (SNHL), attributable to damage (aging, noise exposure, trauma, genetic factors, intravenous antibiotics and chemotherapy agents, etc.) to the cochlea—our hearing organ.
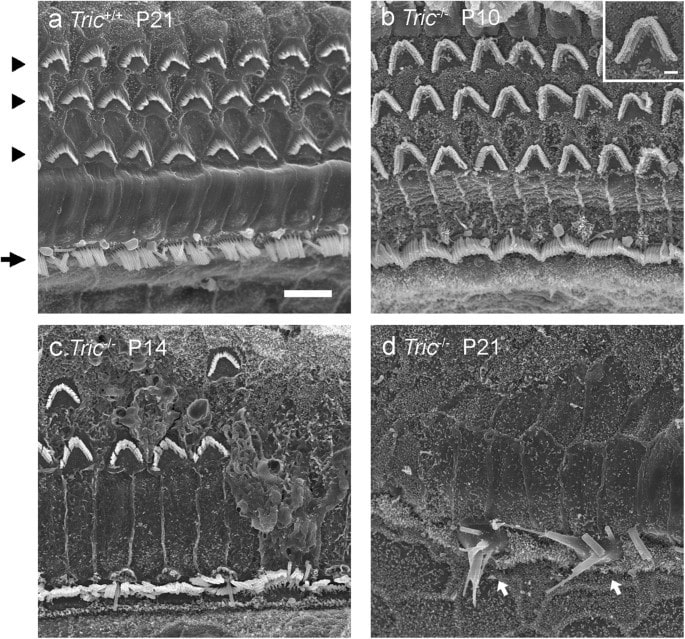
Deletion of Tricellulin Causes Progressive Hearing Loss Associated with Degeneration of Cochlear Hair Cells Toru Kamitani, Hirofumi Sakaguchi, Atsushi Tamura, Takenori Miyashita, Yuji Yamazaki, Reitaro Tokumasu, Ryuhei Inamoto, Ai Matsubara, Nozomu Mori, Yasuo Hisa & Sachiko Tsukita, Scientific Reports volume 5, Article number: 18402 (2015)
Cochlear outer hair cells before (upper 2 photos) and after (lower 2 photos) noise damage
What are the different types of hearing loss?
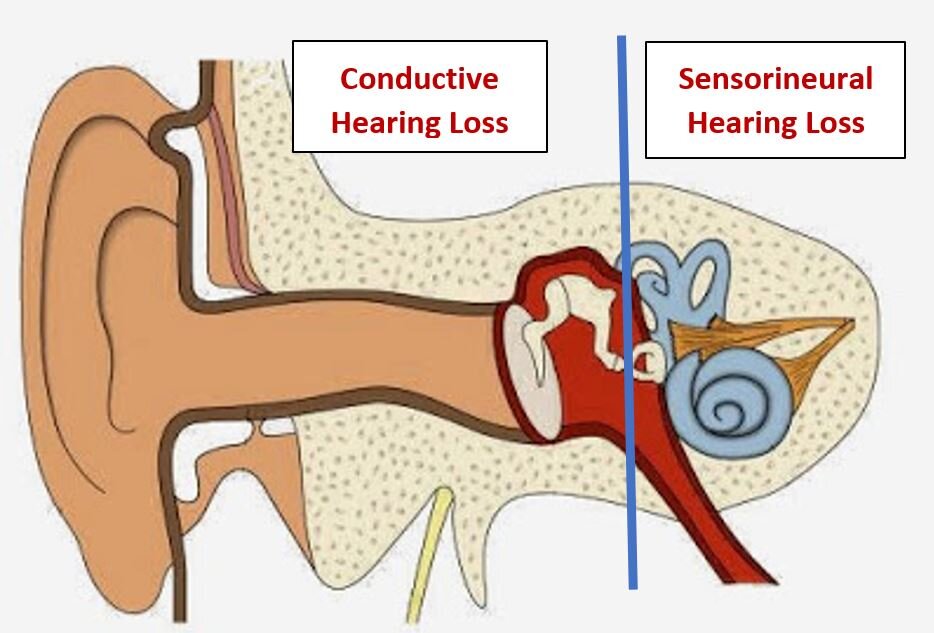
Ear, nose and throat doctors (otolaryngologists) like to measure the degree and type of hearing loss in our patients in order to plan the best treatment. Accordingly, we have divided hearing loss into two main categories: conductive hearing loss and sensorineural hearing loss. One can have a combination of both in each ear. This does not indicate the original causes of the hearing loss but only to the type of mechanical or physiologic damage or deficiency in hearing.
 https://www.ent4kids.co.uk/surgery-information-archive/grommet-insertion
https://www.ent4kids.co.uk/surgery-information-archive/grommet-insertion
We have had excellent and ever-increasing success over the past 100 years treating conductive hearing loss with medication and/or surgery. This type of hearing loss is due to changes, damage, or deformities of the sound conducting mechanisms in the outer or middle ear.
Progress in directly treating the source of sensorineural hearing loss—on the other hand—has been dismal. This type of hearing resides in the cochlea (hearing organ), the hearing (acoustic) nerve, and the brain. Aside from several very special (and unfortunately rather rare) cases, a medication or surgical procedure is not available for improving sensorineural hearing. There are surgical procedures on the cochlea: (the cochlear implant—which replaces the elegant sensory mechanism with a wire array of a few electrodes on a wire), or a surgical procedure bypassing the cochlea completely (the auditory brainstem implant). In certain severe circumstances these procedures are somewhat useful in approximating hearing. They bypass but do not truly repair the damaged natural sensorineural hearing mechanism which is due to nerve and organ damage at the cellular level.
Of course, hearing aids are very useful in correcting most types of hearing loss, but they rely a priori on the cochlea’s ability to decipher speech. In other words, a sophisticated hearing aid of the highest quality is not very effective in helping speech understanding individuals who can only understand 50% of speech in quiet situations—under the best of conditions they will miss one out of every two spoken words.
What causes the loss in understanding speech?
The most common causes of hearing loss are noise exposure and the aging process. Over time these factors damage the cochlea and the result of the damage can be measured on the standard hearing test, the audiogram. This often shows a decrease in hearing in the higher pitches (frequencies) in both ears and a decrease in the ability to hear 2-syllable equal accent words (“ice cream, baseball, cowboy, etc.”, called spondees). These are the commonly measurable effects of hearing loss.
Before this type of hearing loss is occurring, there are even earlier and far more subtle changes in hearing. We refer to these changes as “hidden hearing loss”, because it cannot be measured with standard hearing tests. It results in the inability to “pick out” and understand speech in the presence of noise. This is the first sign that hearing loss is occurring. It can be measured with more sophisticated testing.
Ten years ago, researchers at Harvard Medical School—Massachusetts Eye and Ear Infirmary came up with the first answers regarding what was happening. The aging process and noise exposure causes a loss of synapses (nerve fiber connections) between the sensory cells (inner hair cells) and the hearing nerve (acoustic nerve/spiral ganglion cells) as seen in experimental animals:
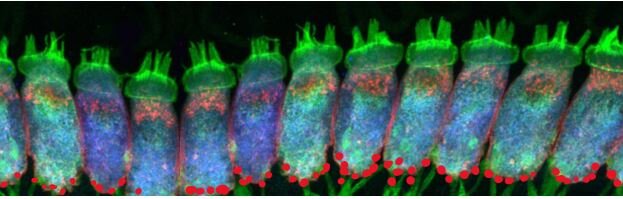
Auditory synaptopathy, auditory neuropathy, and cochlear implantation Aiden Eliot Shearer MD, PhD, Marlan R. Hansen MD, First published: 01 July 2019 https://onlinelibrary.wiley.com/doi/10.1002/lio2.288
Inner hair cells with hairs (green, top), cell bodies (blue), acoustic nerve fibers (green, bottom) and synapses (red,bottom).
This effect is best illustrated in the following simplified diagram, showing the “pruning” of synapses and peripheral terminals (blue arrows) in the cochlea before and after loud noise exposure:
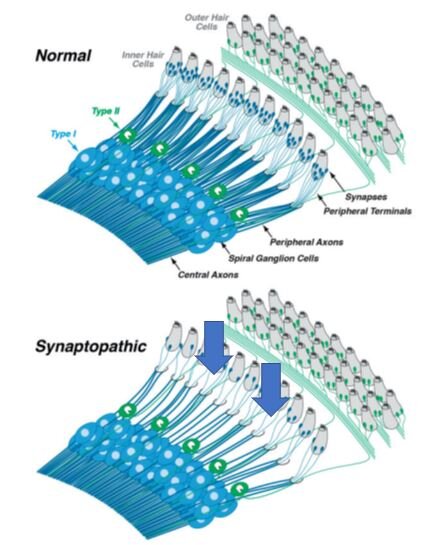 In the normal ear, each inner hair cell sends signals to the brain via numerous type-I spiral ganglion neurons (top schematic). In the synaptopathic ear (bottom schematic) many of the synaptic contacts between type-I (acoustic nerve or spiral ganglion) neurons and inner hair cells have degenerated or have been “pruned” away (blue arrows), thus many of the surviving spiral ganglion cells are silenced. The type-II spiral ganglion neurons (green) innervating outer hair cells are not involved in hearing and are unaffected. The central axons convey the sound information to the brain by way of the acoustic nerve.
In the normal ear, each inner hair cell sends signals to the brain via numerous type-I spiral ganglion neurons (top schematic). In the synaptopathic ear (bottom schematic) many of the synaptic contacts between type-I (acoustic nerve or spiral ganglion) neurons and inner hair cells have degenerated or have been “pruned” away (blue arrows), thus many of the surviving spiral ganglion cells are silenced. The type-II spiral ganglion neurons (green) innervating outer hair cells are not involved in hearing and are unaffected. The central axons convey the sound information to the brain by way of the acoustic nerve.
Similar effects have been detected in the aging cochlea in experimental animals. Therefore, the content and amount of information reaching the brain has likewise been greatly decreased—leading to difficulty in interpreting speech in a noisy environment. Some loss of synapses is thought to occur before any changes measurable by standard audiograms and after as little as 2 hours of exposure to sounds of 90 dB (decibels) or higher. The need to avoid loud noise for prolonged periods cannot be stressed enough!
The bad news:
The loss of synapses and speech discrimination ability is presently irreversible.
The good news:
There is recent research that strongly suggests that a common prescription medication can improve and/or mitigate this type of hearing loss.
What is the possible answer to this problem?
Many elderly women and some elderly men suffer from osteoporosis, a bone-weakening disease. Twenty years ago, it was recognized that a class of drugs called bisphosphonates can reverse this process. These drugs were widely prescribed for this disease and drug research and development yielded two additional generations of this drug class. Eventually, the third-generation bisphosphonates displayed other interesting and therapeutic properties. They were used to control metastatic bone pain. For example, the drug minodronic acid (YM529) has been shown to directly and indirectly prevent proliferation, induce cell death (apoptosis), and inhibit metastasis of various types of cancer cells.
Significantly, some of these patients on third-generation bisphosphonates began to report improvement in their ability to hear and understand speech.
On July 14, 2020, researchers from the original Massachusetts Eye and Ear Infirmary and Eaton-Peabody Laboratories (Konstantina M. Stankovic, MD, PhD, and Albert Edge, PhD, et al.) reported that in animal models a particular bisphosphonate reversed cochlear synaptopathy. In other words, the drug induced repair and regrowth of synapses that were lost due to the noise damage to the cochlea.
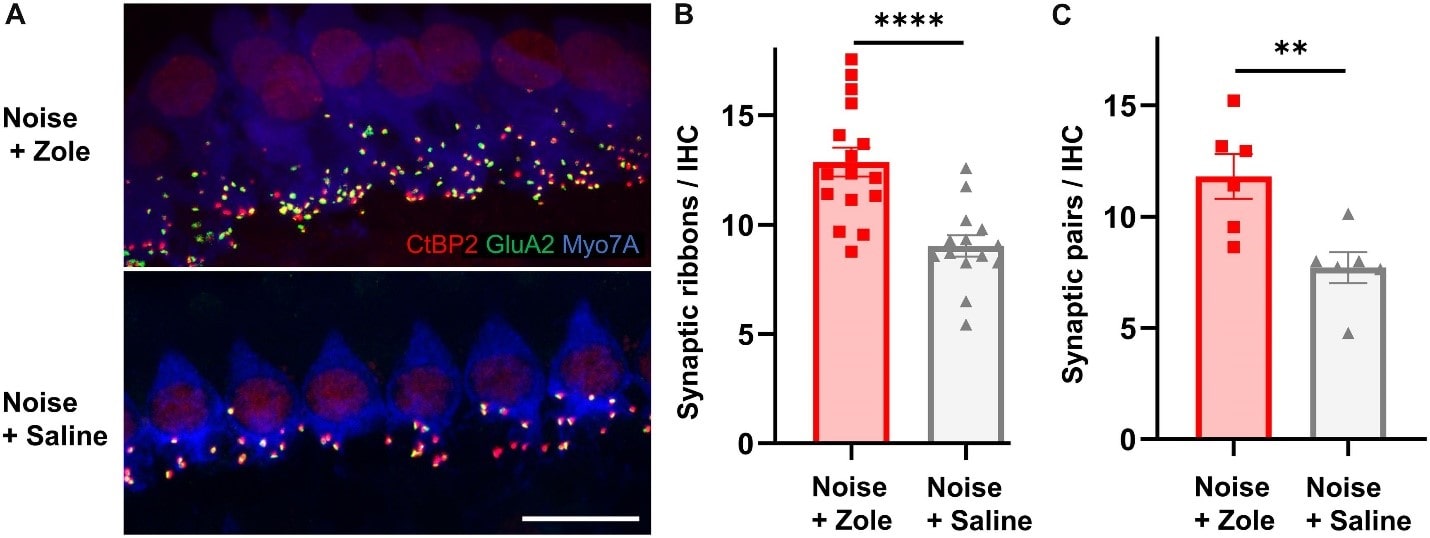
Regeneration of Cochlear Synapses by Systemic Administration of a Bisphosphonate Front. Mol. Neurosci., 14 July 2020 Richard Seist, Mingjie Tong, Lukas D. Landegger, Sasa Vasilijic , Hiroshi Hyakusoku, Sachiyo Katsumi, Charles E. McKenna, Albert S. B. Edge & Konstantina M. Stankovic https://www.frontiersin.org/journals/molecular-neuroscience/articles/10.3389/fnmol.2020.00087/full
The upper photograph shows a larger number of synapses (colorful dots) on noise-damaged inner hair cells treated with the bisphosphonate as compared to the lower photo of noise-damaged inner hair cells not treated with the drug.
This is the first time that a drug has been demonstrated to have a beneficial and reparative effect on the cochlea.
What does the future hold for this problem?
Understanding of the mechanism of hidden hearing loss allows the focus of research to be directed at developing techniques of repairing the synaptic damage. Along those lines, a parallel tract of research is directed at gene therapy to 1) repair specific genes causing sensorineural hearing loss in genetic hearing diseases and 2) toward altering cellular controls to enhance repair of damaged synapses in aging and/or noise-exposed individuals.
The repurposing of bisphosphonates shows promise in animal models for treating hidden hearing loss. At the present time, these drugs are not fit for hearing treatment because they have not yet been validated in human studies. For the purposes of treating metabolic bone disease—osteoporosis, etc.—they have demonstrated success. However, they are associated with some significant side effects and are absolutely contraindicated in pregnancy (they are known to cause birth defects). In time and with proper vetting, these techniques may ultimately be valuable treatments for a very common type of hearing loss.
Find an ENT & Allergy Associates Doctor Near You, or Explore More Blog Topics
