As the world waits for the first effective anti-SARS-CoV-2 (the virus that causes COVID-19) vaccine, we face significant limits on stopping or slowing the pandemic. We have relied on social distancing, quarantine, hand washing, contact tracing, testing, and the use of barriers (masks, respirators, gloves, goggles, gowns, etc.). to prevent transmission of the virus from one individual to another. These measures (aside from testing) have been known for over a hundred years and were used to stop the Spanish Flu pandemic of 1917-1919.
Now researchers from the University of California-San Francisco and the Howard Hughes Medical Institute announced on August 11, 2020 the use of synthetic nanobodies to block the virus from infecting our cells.
What are “nanobodies”?
First discovered in European labs in the 1980’s, nanobodies are antibody fragments that are produced naturally only in certain camelid (camel-like) species such as llamas:

Andean Llamas
In nature and only in camelids, these nanobodies are produced along with conventional antibodies and heavy chain antibodies in response to infection or invasion by bacteria, fungi, allergens, or viruses. Production of these conventional antibodies (which we also produce) is the goal of all anti-COVID-19 vaccines. The advantage of the conventional antibody is that once bound to the invader, they can signal a subset of the T-cells (“killer” white blood cells) to target and destroy the invader.
But they are big, complex molecules that are short-lived outside of the body and difficult to mass-produce. Currently, they are only available by extraction from “convalescent plasma” donated by individuals who have recovered from COVID 19 and therefore in short supply. Conventional antibodies can only be delivered by a health care professional through intravenous injection. Outside the body, they are relatively fragile and very quickly lose potency.
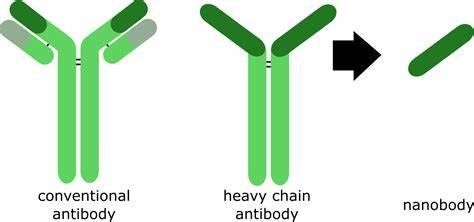
The advantage to nanobodies is that they remain stable for long periods of time outside the body and are just as specific toward the virus as conventional antibodies. In the case of the COVID-19 virus, nanobodies are even more specific than conventional antibodies. Nanobodies can be delivered through a variety of mechanisms (oral, IV, nebulizer, and nasal spray). They are simpler to mass-produce. These nanobodies can be freeze-dried, heat-treated, and aerosolized without damage. The disadvantage of nanobodies is that they have no T-cell-signaling capability. Nonetheless, when properly configured, they are capable of completely interfering with viral invasion and replication processes. They can be produced in large quantities in a relatively short period of time, according to researchers.
How does the coronavirus SARS-CoV-2 infect us?
The virus causing COVID-19 infects us through entry into our mucous membranes, typically incubating for several days in our nose and nasopharynx. This is the reason for nasal swab testing in the COVID-19 tests and the probable reason for the early infection symptom of loss of smell. Hence, the interest in this mechanism by ENT doctors.
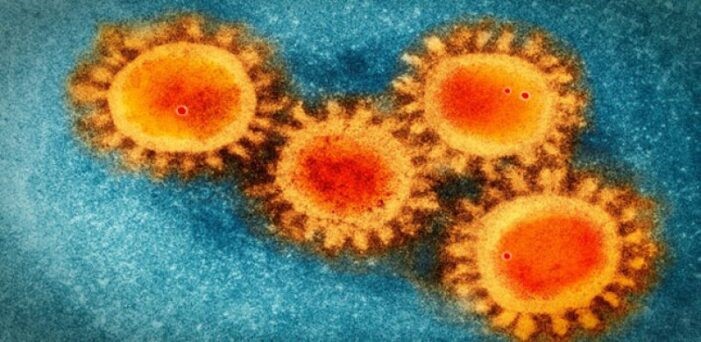
Once in our bodies, the virus must enter our cells and commandeer the cellular mechanisms in order to reproduce. To do this, it must enter our cells by using a “spike protein” which contains a binding site for the human ACE2 (acetylcholine esterase 2) enzyme, found in abundance on the surface of most of our cells. (Please see my previous blog: “Face to Face with “Long-Haul” COVID-19”). About 20 spike proteins are on the surface of the virus, giving it a “corona-like” appearance:
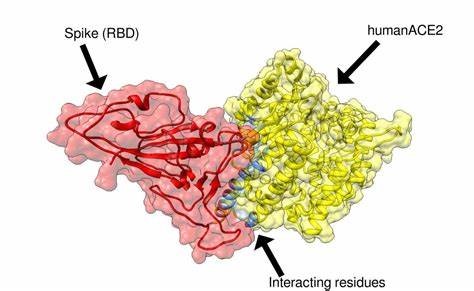
The interaction between the spike protein on the surface and the ACE2 protein on our cell surfaces involves a series of steps that is understood to occur in a special sequence:
- The spike binding area physically opens-in a hinge-like manner- revealing the "receptor binding domain" (RBD)
- The now-exposed RBD has great affinity for the human ACE2 protein and binds to it, "lock and key" fashion.
- The binding induces the viral genetic material to enter the cell and to start the production of multiple virus copies, which ultimately destroys the cell releasing many more virus particles.
How do nanobodies work?
Using the technique of “cryogenic electron microscopy”, a unique nanobody was identified (Nb6) by the University of California at San Francisco (UCSF) group that binds very strongly to the spike RBD, thus interfering with the spike’s ability to bind to the human ACE2 receptor (Step 2 above).
In addition to this, Nb6 was shown to be able to “cap” or prevent opening of the hinge mechanism that is necessary to expose the RBD to the ACE2 receptor (Step 1 above. No RBD, no ACE2 binding, no infection. The virus cannot multiply without entering our cells!
Furthermore, it appears that Nb6 can accomplish this in infinitesimally small concentrations. If one links 3 Nb6’s together (termed mNb6-tri) in a chain, even greater stability and affinity for viral binding exists than the single Nb6.
This permits reliable stability in solution and allows for liquid or aerosolized preparations. It is this stability and ease of use that differentiates it from the more common monoclonal antibodies that are in ever widening use in medicine today.
What does this have to do with ENT?
One of the first signs of COVID-19 infection in many people is the loss of sense of smell (anosmia). At this early point patients can be tested. If COVID-19 positive, patient treatment with nanobodies can begin to block further COVID-19 expression. This can be delivered via nebulizer or nasal spray to the upper airway. In fact, the researchers at UCSF have termed the aerosolized nanobodies “AeroNabs” and are actively seeking to team up with manufacturers and begin clinical trials shortly. These preparations would be surface-active and could be used upon suspected exposure to COVID-19 or during treatment of the disease. The ultimate weapon against COVID-19 will be an effective vaccine, but until that day, these nanobodies offer hope of decreasing death and infection rates.
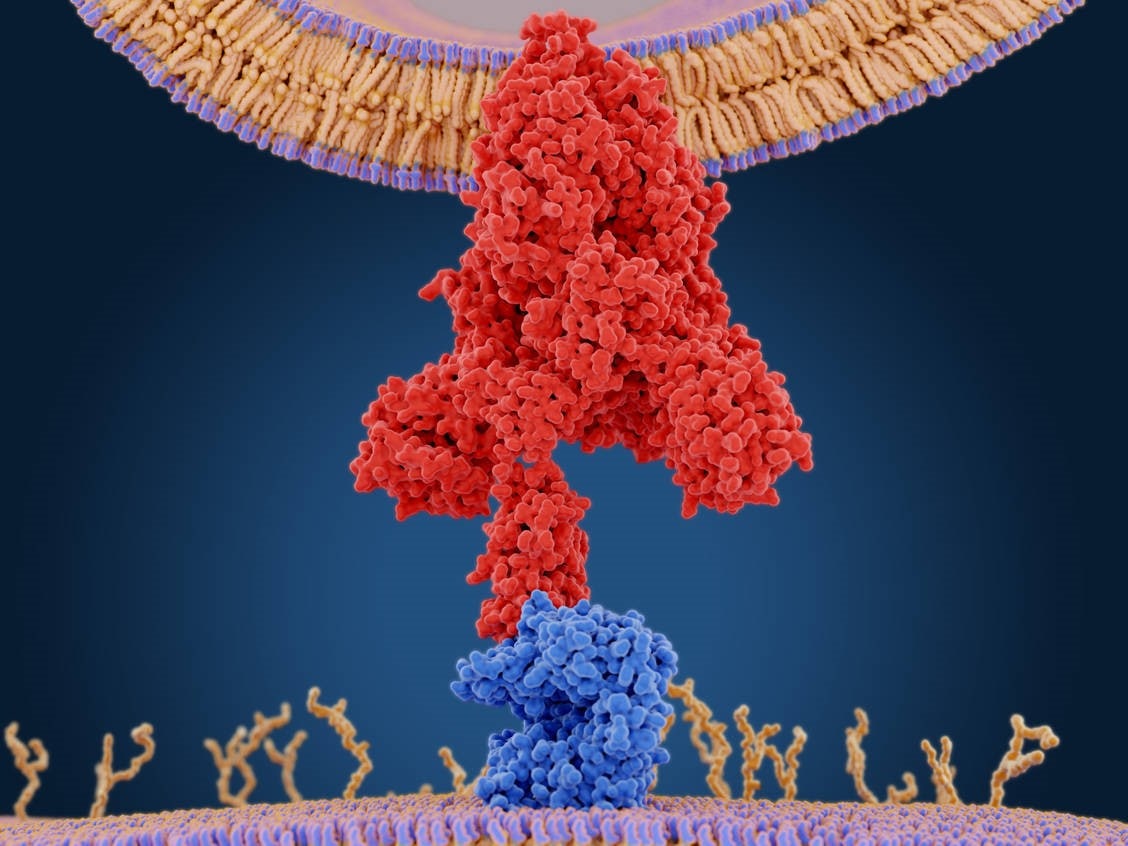
Schematic of SARS-CoV-2 Spike Protein (Red) Binding to the Human ACE-2 Protein (Blue)
Ref: https://www.biorxiv.org/content/10.1101/2020.08.08.238469v1
Find an ENT & Allergy Associates Doctor Near You, or Explore More Blog Topics
